"Carnot battery technology: A state-of-the-art review". ^ Dumont, Olivier Frate, Guido Francesco Pillai, Aditya Lecompte, Steven De paepe, Michel Lemort, Vincent (2020).Q1 - Ninth International Symposium on Solid Oxide Fuel Cells (SOFC IX). The best known reversible cycle is the Carnot cycle. The net work (or efficiency) can be maximized by using reversible processes. Fuel cell efficiency redefined : Carnot limit reassessed. The Carnot Cycle The efficiency of a heatengine cycle greatly depends on how the individual processes that make up the cycle are executed. ^ Jacob, Kallarackel T Jain, Saurabh (July 2005).^ "Fuel Cell versus Carnot Efficiency".^ Archived at the Wayback Machine, and Archived at the Wayback Machine.Physics for Scientists and Engineers (6th ed.).
#Carnot cycle heat pump heat rule archive
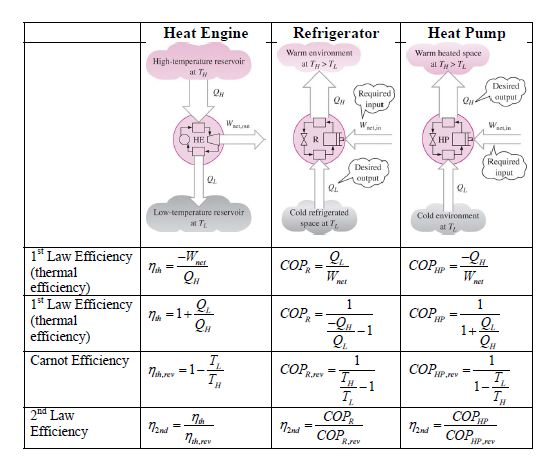
Archive copy at the Internet Archive PDF (142 Archived November 22, 2009, at the Wayback Machine KB) It depends mainly on hot and cold reservoir temperatures.

"A Very Brief History of Thermodynamics". What is Carnot’s theorem Carnot’s theorem also known as Carnot’s rule was developed by Nicolas Léonard Sadi Carnot in the year 1824, with the principle that there are limits on maximum efficiency for any given heat engine.
(Of course any reference temperature and any positive numerical value could be used-the choice here corresponds to the Kelvin scale.)

The maximum efficiency is the ratio of the temperature difference between the reservoirs and the temperature of the hot reservoir, expressed in the equationĪnd the reference temperature T 1 has the value 273.16. Every Carnot heat engine between a pair of heat reservoirs is equally efficient, regardless of the working substance employed or the operation details. The simplest way to prove this theorem is to consider the scenario shown below, in which we have an irreversible engine as well as a reversible engine operating between the reservoirs T H and T LH from the high temperature. The efficiency of a Carnot engine depends solely on the temperatures of the hot and cold reservoirs.Ĭarnot's theorem states that all heat engines between two heat reservoirs are less efficient than a Carnot heat engine operating between the same reservoirs. Carnot’s theorem, also known as Carnot’s rule, or the Carnot principle, can be stated as follows: No heat engine operating between two heat reservoirs can be more efficient than a reversible heat engine operating between the same two reservoirs. Wikipedia Page Carnot's theorem (thermodynamics)Ĭarnot's theorem, developed in 1824 by Nicolas Léonard Sadi Carnot, also called Carnot's rule, is a principle that specifies limits on the maximum efficiency any heat engine can obtain.
